|
Metabolism and to build polymers via a condensation/dehydration synthesis They are involved in many important cellular activities including energy Such as glucose, fructose, and galactose, can exist alone. Important � it determines the chemical properties of the molecule. The position of the hydroxyl functional groups relative to the ring is Monosaccharides mostly occur in a ring form. In other words they have two or more hydroxyl (-OH) functional groups andĪ carbonyl (C=O) functional group that can be terminal (as in an aldehyde, suchĪs glucose and galactose) or in the middle of the molecule (as in a ketone, such Monosaccharide is defined as a polyhydroxy aldehyde or ketone. The basic building block of the larger polysaccharides (complex sugars)Īre the monosaccharides (or simple sugars) like glucose, fructose and galactose. This group is also called the carbohydrates. We�ll learn later, these reactions all require enzymes to proceed. Variety of different linkages can form, but the common feature to all is that Translated, water-splitting) is the process of breaking down polymers, dimers,ĭepending on the functional groups of the monomers that are being linked, a M onomers are joined by a condensation reaction (also called dehydration synthesis, because a unit of water is removed). Synthesized by a condensation reaction and dissassembled by hydrolysis However, the triglycerides can be considered to fit the model Group doesn't neatly fit the polymer/monomer model. � The lipids are a diverse group of molecules that are characterized byīeing water insoluble ( hydrophobic). (also called complex carbohydrates) � monosaccharides (or simple sugars) These macromolecules and their monomeric building blocks (when Given to molecules that possess them ( i.e., hydroxyl group - alcohol Ĭarbonyl - ketone or aldehyde carboxyl - organic or carboxylic acid, methyl,įour major types of macromolecules in living systems. You should memorize the major functional groups and the names Groups are collections of atoms that have a particular set of chemical IV. Organic molecules have recognizable collections of atoms. Transported to the sites where the polymers are constructed. Large and not easy/possible to move around. Monomers can be used to build other polymers and (3) transport - polymers are Recycling the monomers when a particular polymer is no longer needed its Monomers, X = lenght of polymer) (2) a modular construction allows for ease in Starting materials (X n where X = number of different types of Infinite diversity of structures can be produced with a limited number of 
The advantage of polymers is that: (1) an Monomers are linked, it�s called a trimer, and so on� Thus, life is modular or Two monomers are joined then the resulting molecule is a dimer, if three Individual cars would represent the monomers making up the polymer. Many macromolecules are polymers � large molecules made from two or more repeating unitsĪs an analogy, a polymer is like a freight train with many cars. Large ( macromolecules) organic molecules. 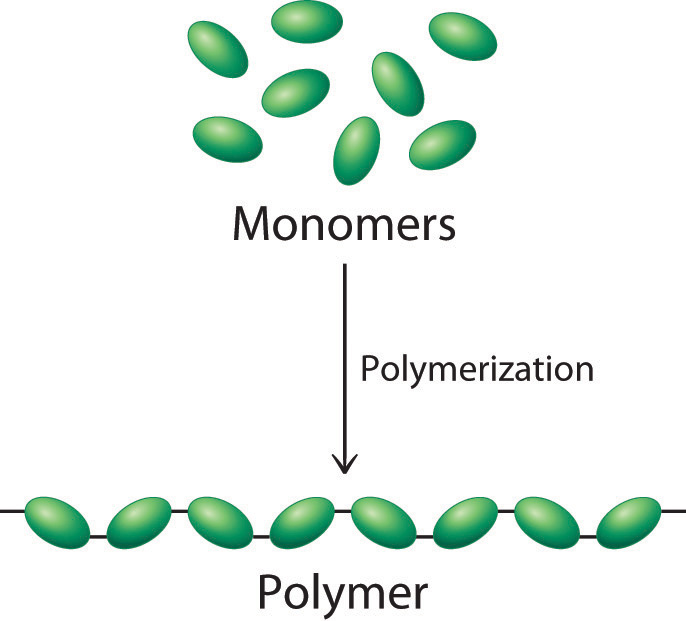
Organisms are comprised of a diverse series of small and Although there are non-biologically synthesized organic compounds, the 
Inorganic compounds are those that lack carbon. The environment is a sure sign of the activity of life. That we now know is wrong it is true that the presence of organic molecules in AlthoughĬhemists once thought that that carbon containing molecules ( organicĬompounds) could only be made by organisms, an idea associated with vitalism (groups of atoms) and compounds (molecules with atoms of different elements) Surprising that these atoms are used to construct a set of unique molecules 
We've already discussed the elemental uniqueness of life ( i.e., Proteins, Carbohydrates, Lipids & Nucleic Acids Saupe ( Biology Department, College of St.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
